 
The state of that system at a particular time is given by the wave function, a solution to this equation, which can only give probabilities for the values of particular properties of the system. A quantum system, such as the electron Heisenberg was thinking of, can be described mathematically using Schrödinger's equation. The uncertainty captured in the principle is inherent in the quantum world, whether it is observed or not.Īlthough Heisenberg's uncertainty principle might seem a confusing idea, it has a simple clarity when you describe the situation mathematically. "Heisenberg's uncertainty principle is often phrased in terms of measurement, but really you should think the electron itself," says Ben Allanach, professor of theoretical physics at the University of Cambridge. Heisenberg's thought experiment might seem clear but is misleading as it could suggest that the uncertainty principle is a result only of the disturbance caused by observation (this is another phenomenon, known as the observer effect). So a gamma-ray microscope would give you a highly accurate measurement of an electron's position, but the disturbance would create a less accurate measurement of the momentum. But the energy of gamma-ray photons is so high, the collision would affect the motion, and hence the momentum, of the electron. ) Heisenberg reasoned you could very accurately measure an electron's position with a gamma-ray microscope, but to do so you have to bounce at least one gamma-ray photon off the electron. And the energy,, of a photon is given by. 
The speed of light is fixed at so as the wavelength decreases the frequency must increase. (This is because for waves, where is the speed of the wave, is the wavelength and is the frequency. Heisenberg suggested using a gamma-ray microscope as gamma-rays are a type of light with a very short wavelength.īut the tradeoff is that light waves with a shorter wavelength have a higher frequency, and the photons have correspondingly higher energy. The accuracy of a microscope is limited by the wavelength of the light used – the shorter the wavelength the more accurate your observations. To do this you would use a microscope which bounces photons of light of an object in order to observe it. Suppose you wanted to locate the position of an electron. Heisenberg himself tried to understand this principle physically using the following thought experiment. But the principle extends to other pairs of particles (called complementary variables), such as length of time and energy, whose connection is expressed in similar inequalities. This is the most common mathematical expression of Heisenberg's uncertainty principle, that the product of these uncertainties has a minimum value. Where is the Planck's constant (a kind of order of magnitude of the quantum world) divided by. If we write the uncertainty in a particle's position as, and the uncertainty in a particle's momentum as, then the uncertainty of these two properties is connected in the following way: (The momentum of an object is its mass times its velocity.) The uncertainty principle states that you cannot know, with absolute certainty, both the position and momentum of an electron – the more accurately you measure one of these properties the less accurate your knowledge of the other. In 1927 the German physicist, Werner Heisenberg, framed the principle in terms of measuring the position and momentum of a quantum particle, say of an electron. 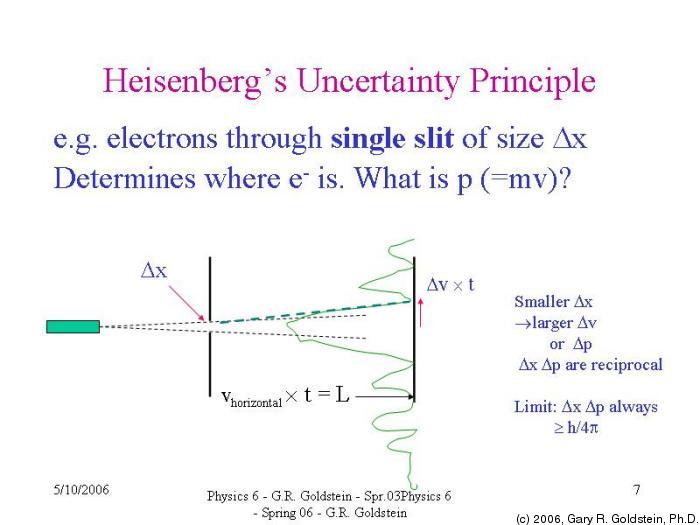
This quantum fuzziness lies behind one of the most famous principles of quantum physics: Heisenberg's uncertainty principle. Werner Heisenberg in 1933 (photo: German Federal Archive) 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
